CELL THERAPY
NestaCell® - Registered Trademark - Patent Granted in USA and Europe
NestaCell® product cells are produced in a controlled "ISO5" environment surrounded by ISO6 and ISO7 controlled environments.
Cellavita facilities and Good Manufacturing Practices for Advanced Cell Therapy products follow the regulations of RDC 441/2011, RDC 11/2009 and part of RDC 55/2015, in addition to the specific requirements of RDC No. 508, of May 27, 2021, on Good Practices in Human Cells for Therapeutic Use and clinical research.
NestaCell® product quality and production parameters follow Anvisa and Brazilian Pharmacopoeia regulations, FDA (Food and Drug Administration), International Society for Cell Therapy and EMA (European Medicines Agency).
Other Cellavita Facilities
- Quality control
- Storage
- Cryopreservation room
- Offices
- Quality Assurance
- Washing and waste disposal room
- Materials and biological material receiving room
Approval of Studies in Humans
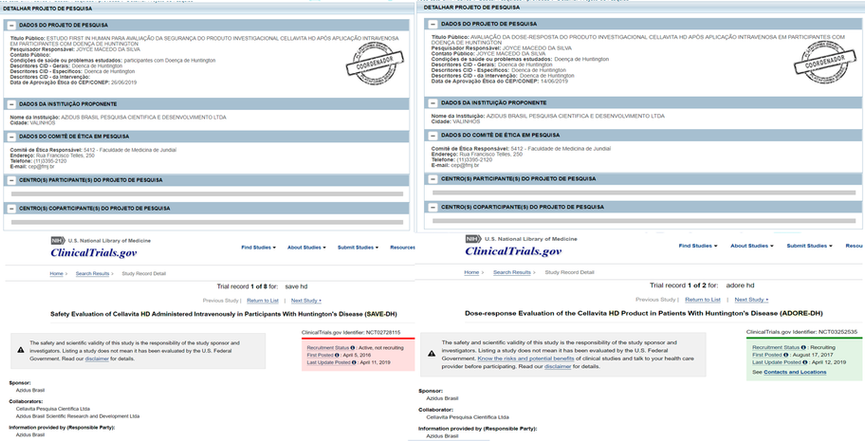
ClinicalTrials.gov - Cellavita Studies
Project Approved by FINEP

