COVID-19 PANDEMIC
Phase II Clinical Study (pilot according to WHO)
Evaluation of the Efficacy and Safety of NestaCell® Mesenchymal Stem Cells in the treatment of hospitalized patients infected with the SARS-CoV-2 virus (COVID-19).
World Scientific Literature:

Pre-Clinical Biodistribution Studies:
NestaCell® product behavior in vivo
Biodistribution capacity of NestaCell® product cells in vivo after intravenous administration. After 4 hours, the BLM signal of labeled cells was detected in the lung in 100% of animals in the lung region. Cells remained in the organ (lung) for up to 30 days.
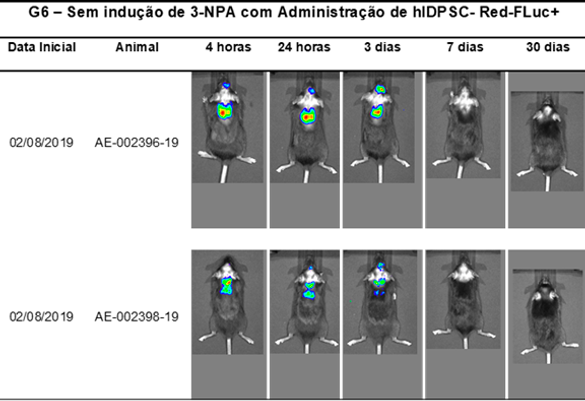
Bioluminescence images of luciferase-labeled cells (Hidpsc- Red-Luc+) from C57 black lineage animals in ventral decubitus, acquired with IVIS Lumina II (Xenogen Corp. CA, USA).
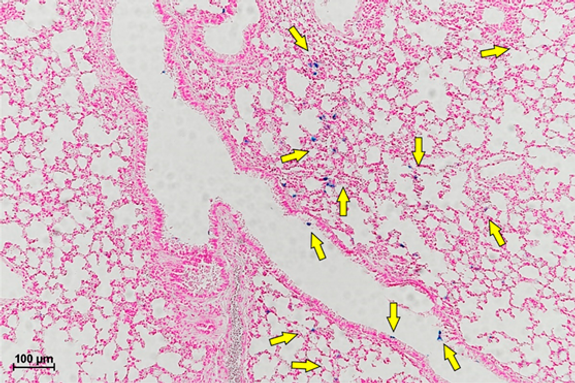
Optical microscopy images of lung tissue using Fast Red and Prussian Blue staining, group G4, at 30 days.
Primary Efficacy Objective of the Study and its variable
To evaluate the efficacy of advanced cell therapy NestaCell® in promoting reduction in the length of stay on invasive mechanical ventilation in hospitalized COVID-19 patients.
Participants' clinical status throughout the study will be assessed by the Ordinal Scale for Clinical Evolution Assessment. Grades 6 and 7 on this scale correspond to invasive mechanical ventilation.
Study Design
This Phase 2 clinical trial will be prospective, double-blind, randomized, parallel, interventional, multicenter and placebo-controlled.
Treatment Groups
90 participants will be included in the study, randomized in a 1:1 ratio to the Test (NestaCell®) or Control (placebo, NestaCell® vehicle) groups. 45 participants in each group.
- Test Group: NestaCell®
- Control Group: Placebo (investigational product vehicle)
Investigators and Main Research Centers
- Dr. Florentino Cardoso – Oncologist and Intensivist – Casa de Saúde de Campinas
- Dr. Ricardo Diaz – Infectious Disease Specialist – Escola Paulista de Medicina (Unifesp)
- Dr. Alexandre de Matos Soeiro – Cardiologist and Intensivist – Incor (HC-USP)
- Dr. Paulo Fernando Guimarães Morando Marzocchi Tierno – Intensivist – SPDM (São Paulo)
CONEP Approval